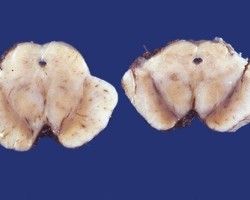
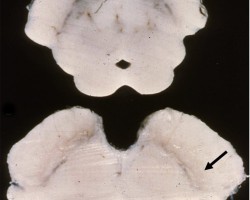
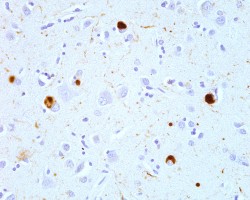
In this chapter, we will cover the typical semiology of hypoactive and hyperactive movement disorders as well as discuss their treatments. The imaging, pathology, and genetics within this chapter are especially important: review the text and test your knowledge with a quiz at the end.
Authors: Eric Jackowiak MD, Brian Hanrahan MD, Robyn Massa MD, Steven Gangloff MD
Editor: Yasmin Aziz MD
Hypoactive Movement Disorders
- Hypoactive movement disorders are due to reduced activity of the direct pathway.
- D1 dopamine receptors are the main dopamine receptor in the direct pathway.
- See our Basal Ganglia and Cerebellum chapter for more details about the direct pathway.
Reminder
D2 receptors are preferentially located in the mesolimbic and mesocortical pathways
Parkinson’s Disease (PD)
- Presents with unilateral resting tremor, cogwheel rigidity, bradykinesia, and hypomimia.
- REM sleep behavior disorder, constipation, and anosmia can present years before disease onset.
- REM sleep disorder: Complex nocturnal behaviors involving vocalizations, hitting, punching, or gesturing.
- Major depression can be seen in half of patients.
- Later non-motor features include autonomic dysfunction and hallucinations.
- PD with dementia: Diagnosis of PD for at least 1 year before the onset of dementia symptoms.
- 10 to 15% of patients with PD have a familial form:
- LRRK2 mutations (autosomal dominant) lead to 10% of familial and 5% of sporadic PD cases. It is seen more often in North African Arabs.
- PARK1 gene (α-synuclein) and PARK2 (Parkin) mutations are also possible causes of familial PD.
- Pathology: Alpha-synuclein inclusions/Lewy bodies primarily within the substantia nigra and locus coeruleus.
- Diagnostic studies:
- Diagnosis is most often made based on clinical history and examination.
- In select cases, dopamine transporter (DAT-SPECT) scans may be done. This measures the availability of striatal dopamine.
- DAT imaging would be normal in patients with essential or drug-induced tremors.
- PET scans for 11F or 11C dopa measure dopa decarboxylase activity while PET scans for 11C DTBZ assess vesicular monoamine transporter-2 activity.
Parkinson’s Therapies
PD therapies are aimed at increasing intracranial dopamine effects by stimulating its production (dopamine precursors), mimicking its action at receptors (direct agonists), blocking its peripheral conversion (carbidopa), preventing inactivation (COMT inhibitors), or decreasing its breakdown (MAO-B inhibitors).
Dopamine precursor (Levodopa)
- Mechanism of action: Levodopa is converted into dopamine after it crosses the blood-brain barrier thus increasing intraparenchymal levels of dopamine.
- Side effects: Nausea/vomiting, dyskinesias, orthostatic hypotension.
- Carbidopa is given concurrently with levodopa to reduce the GI side effects and reduce levodopa’s peripheral plasma breakdown.
- Levodopa has a short half-life which can lead to motor fluctuations and peak-dose dyskinesia.
- Levodopa is safe in pregnancy.
COMT inhibitors (tolcapone and entacapone)
- Mechanism of action: Reduces methylation of levodopa and dopamine, which increases levodopa’s half-life.
- Ineffective when given alone but can prolong the duration of clinical response of levodopa when given together.
- The use of tolcapone requires liver enzyme monitoring.
- Side effects: Severe diarrhea and discoloration of urine.
MAO-B inhibitors (rasagiline and selegiline)
- Mechanism of action: Decreases the catabolism of dopamine, resulting in a greater peak effect and less wearing off when used in combination with levodopa.
- Can also be used as monotherapy in early PD.
- Side effects:
- Ingestion of tyramine heavy foods (aged cheeses, cured meats) can result in severe tachycardia and hypertensive crisis which at times can be fatal.
- Concurrent use of SSRIs can lead to serotonin syndrome.
- Decongestant medications and some narcotics may also interact with MAO-B inhibitors.
Direct dopamine agonists (DDA)
- Bromocriptine, pramipexole, ropinirole, rotigotine, and apomorphine
- Side effects: Impulse-control disorder, hallucinations, nausea, orthostatic hypotension, peripheral edema.
- Pramipexole and ropinirole have been associated with excessive drowsiness and falling asleep while driving.
- Can worsen dyskinesias when added to levodopa.
- Less likely to cause augmentation than levodopa.
Partial dopamine agonist and partial NMDA receptor antagonist (Amantadine)
- Mechanism of action: Has an anti-dyskinetic effect by reducing the frequency of abnormal involuntary movements due to levodopa.
- Side effects: Livedo reticularis
Anticholinergics (Trihexyphenidyl and benztropine)
- Can be used in younger patients (under 60) with tremor-predominant dysfunction, bradykinesia, postural instability, and/or rigidity.
- Side effects: Cognitive dysfunction, urinary retention, dry mouth, and GI disturbance.
Psychosis-related therapies
- Atypical antipsychotics
- Clozapine
- Antipsychotic of choice for PD-related psychosis.
- Mechanism of action: preferential inhibition of dopamine receptors in the frontal lobe and not the basal ganglia.
- Side effects: bone marrow suppression
- Requires blood count monitoring as a result
- Quetiapine
- Doesn’t have the risk of bone marrow suppression like clozapine but is likely not as efficacious.
- Clozapine
- Pimavanserin
- Only FDA approved medication specifically for Parkinson’s disease psychosis.
- Mechanism of action: Serotonin 5-HT2A receptor inverse agonist and antagonist.
Deep Brain Stimulation (DBS)
- Used to treat the motor symptoms of PD but has no effect on its non-motor symptoms.
- Can be implanted into the contralateral side of major symptoms, or bilaterally.
- Significant cognitive dysfunction is a contraindication to DBS.
- Locations for DBS placement:
- Ventral intermediate nucleus (VIM) of the thalamus: Improves tremor
- Globus pallidus interna (GPi) or subthalamic nucleus (STN) Improves tremor, bradykinesia, and rigidity.
Log in to View the Remaining 60-90% of Page Content!
New here? Get started!
(Or, click here to learn about our institution/group pricing)1 Month Plan
Full Access Subscription
$142.49
$
94
99
1 Month -
Access to full question bank
-
Access to all flashcards
-
Access to all chapters & site content
3 Month Plan
Full Access Subscription
$224.98
$
144
97
3 Months -
Access to full question bank
-
Access to all flashcards
-
Access to all chapters & site content
1 Year Plan
Full Access Subscription
$538.47
$
338
98
1 Year -
Access to full question bank
-
Access to all flashcards
-
Access to all chapters & site content
Popular
Loading table of contents...
Loading table of contents...